The Era of AKI Biomarkers
Consensus to start rethinking the diagnosis of acute kidney injury
Acute kidney injury (AKI) is featured by acute deterioration in renal function, defined by changes in serum creatinine (SCr), urine output, or both. As one syndrome, AKI encompasses a wide variety of causes and risk factors, leading to a decline in kidney function or renal damage. Sepsis, shock, advancing age, cardiac surgery, nephrotoxic medication and heart failure are major risk factors for AKI representing a global burden with 13 million cases per year, and around 2 million deaths (1). Besides, AKI survivors are at higher risk for chronic kidney disease, recurrent AKI, and end-stage renal disease.
Based on the 2012 published Kidney Disease: Improving Global Outcomes (KDIGO) guidelines, diagnosis of AKI and stratification of its severity depends on the magnitude of increase in serum creatinine, and the decrease in urine output (2).
Given the multiple factors (muscle mass, protein intake, gender, age) that affect the concentration of serum creatinine and its slow kinetics the medical community is considering serum creatinine rather as an imperfect gold standard. Especially, in acute care settings, where kidney function changes dynamically, a diagnostic tool without those limitations is still missing (2).
Low urine output is usually a symptom of renal failure but not only it is easily influenced by the use of diuretics and hemodynamic status but it is also non-indicative to the extent of renal deterioration.
Considering the limitations of the current markers in the KDIGO guidelines, new biomarkers are suggested to improve the clinical management of AKI patients. In the past decade, several novel biomarkers have emerged and extensively investigated in clinical trials. Therefore, the 23rd Acute Disease Quality Initiative (ADQI) meeting, was organized to review the current evidence for AKI biomarkers and to conclude recommendations regarding AKI clinical management for clinicians and researchers (2).
From 31 representative publications, the ADQI working group drafted recommendations, which were classified by 4 topics:
- AKI risk assessment
- AKI prediction and prevention
- AKI diagnosis, etiology, and management and
- AKI progression and kidney recovery
AKI risk assessment
Most AKI cases already occur even before the patient presents symptoms of acute illness. For AKI risk assessment, the consensus group suggests to run a Kidney Health Assessment (KHA) that should reveal AKI susceptibility and includes among other factors AKI history, chronic kidney disease and the exposure to nephrotoxic medications. The additional use of damage or functional biomarkers based on the results of the KHA is strongly recommended (2).
AKI prediction and prevention
Considering up to one-third of the AKI cases are preventable, the tools for AKI prediction and prevention will benefit clinical outcome. Along with the clinical assessment validated biomarkers are strongly recommended to triage patients and to optimize treatments. Both, negative and positive results are considered valuable supporting rule-in as well as rule-out decisions (2).
AKI diagnosis, etiology, and management
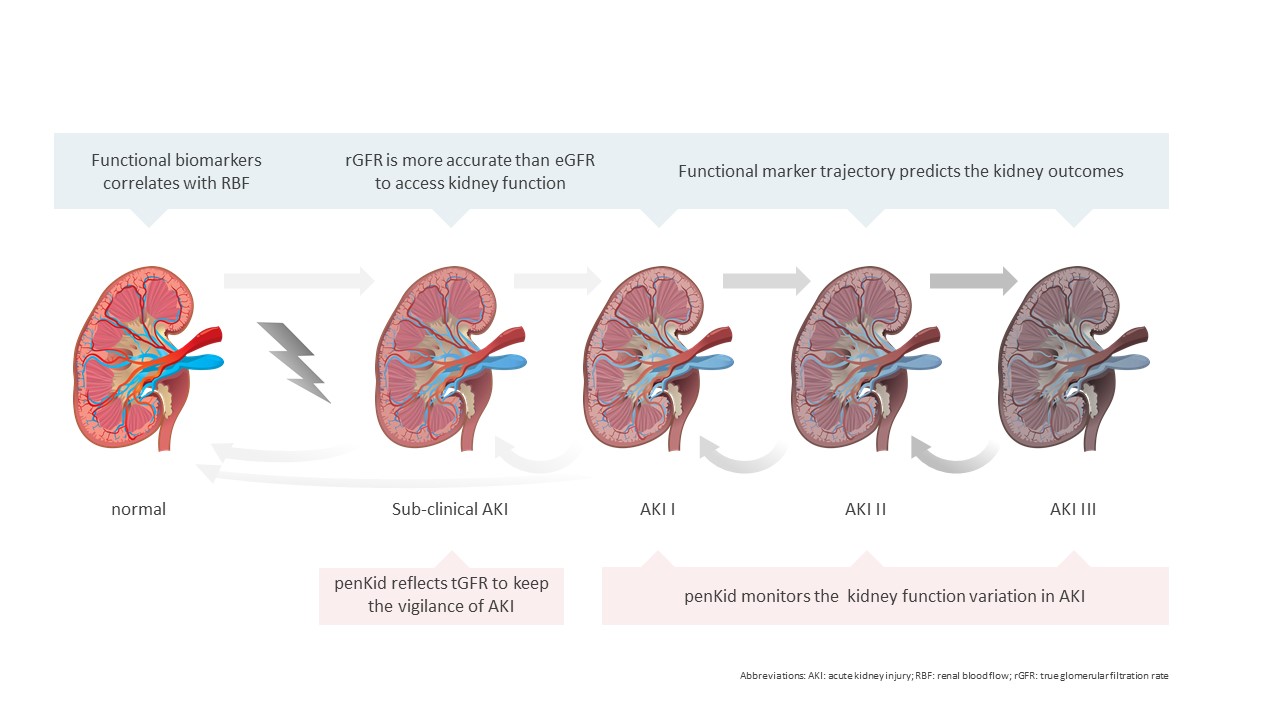
Early identification of patients with AKI is one of the most critical unmet needs of physicians in their daily clinical practice. Based on the high potency of biomarkers in the enrichment of clinical information, the expert panel suggests a modification of the KDIGO stages. Stage 1 AKI can be stratified as two substages to improve the diagnostic accuracy of AKI by the application of biomarkers. The substages are categorized by either both, serum creatinine and damage biomarkers are positive or only one of the criteria is positive. Patients with a positive damage biomarker but a negative serum creatinine may be diagnosed with subclinical AKI- shedding light in the creatinine-blind window, providing an aid in the planning of therapy and management of AKI patients (Figure 1) (2).
AKI progression and kidney recovery
Transient AKI has better clinical outcomes than the persistent AKI, which is defined as the AKI period longer than 48 hours. To better understand the AKI patient’s progression, novel biomarkers are suggested to aid in the prediction (2).
Damage and Functional Biomarkers
AKI typically does not occur as an isolated clinical picture, but rather as a symptom of another event (such as shock or hypovolemia). The cause may be pre-renal, intra-renal or post-renal.
About 60% of the cases are pre-renal and caused are by conditions that result in low kidney perfusion such as low fluid intake or a reduced blood volume due to sepsis or heart failure (3). This initially leads to reduced kidney function and but is not necessarily resulting in tubular damage, only if the condition remains for a long period of time. Intra-renal AKI occurs in about 35% of cases. Toxic medications or bacterial infections can cause direct damage to the kidneys, destroying nephrons, the functional unit filtrating the blood. About 5% of AKI cases are caused by post-renal conditions such as obstructions resulting from kidney stones or a tumor (4).
Though about 20 damage biomarkers are in the pipeline of clinical research, biomarkers for the assessment of functional impairment are limited to serum creatinine, Cystatin C and proenkephalin.
Generally, AKI cannot be linked to specific symptoms and standard diagnostics are failing in acute care settings due to slow changes in such as dynamic condition. The recommended inclusion of new biomarkers in the assessment of AKI is a big step ahead to improve patient management and to start rethinking the definition of AKI.
References:
(1) Farrar A. Acute Kidney Injury. Nurs Clin North Am. 2018 Dec;53(4):499-510. doi: 10.1016/j.cnur.2018.07.001. Epub 2018 Oct 11. PMID: 30388976.
(2) Ostermann M, Bellomo R, Burdmann EA, Doi K, Endre ZH et al; Conference Participants. Controversies in acute kidney injury: conclusions from a kidney disease: Improving Global Outcomes (KDIGO) Conference. Kidney Int. 2020 Aug;98(2):294-309. doi: 10.1016/j.kint.2020.04.020. Epub 2020 Apr 26. PMID: 32709292.
(3) Gameiro, J., Lopes, J.A. Complete blood count in acute kidney injury prediction: a narrative review. Ann. Intensive Care 9, 87 (2019). https://doi.org/10.1186/s13613-019-0561-4
(4) Jonathan S Chávez-Iñiguez, Goretty J Navarro-Gallardo, Ramón Medina-González, Luz Alcantar-Vallin, Guillermo García-García, "Acute Kidney Injury Caused by Obstructive Nephropathy", International Journal of Nephrology, vol. 2020, Article ID 8846622, 10 pages, 2020. doi.org/10.1155/2020/8846622
Disclaimer
The information presented on this blog is meant for health care professionals only. The information provided does not claim to be exhaustive, and is not intended to serve as medical advice, substitute for a doctor’s appointment or be used for diagnosing or treating a disease. For more information please see our Legal Information.